Activated carbon [7440-44-0] Activated carbon has strong electrical conductivity properties. It is an important component of electrodes used in various devices, including torch cells (batteries). Amorphous carbon has some superconducting properties. Graphite is used for “lead” in pencils, as a dry grease and as electrodes in arc lamps. Carbon is an excellent reducing agent because it easily combines with oxygen to form CO and CO2. In this way, in the form of coke in blast furnaces, it cleans metals by removing oxides and other impurities from iron.
Melting point: 3550 ° C
Bulk density: 150-440kg / m3
Cu (copper): ≤0.0025%
Pb (lead): ≤0.001%
Zn (zinc): ≤0.0025%
Residual solvents (Ph Eur / ICH): Excluded from the production process
Sulphated ash: ≤5.0%
Loss on drying (120 ° C): ≤15%
Microbiological purity (aerobic bacteria): ≤1E + 03CFU / g
Microbiological purity (yeast and molds): ≤1E + 02CFU / g
Hazard pictograms
Labeling of hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosive effect, the exclamation mark pictogram should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms




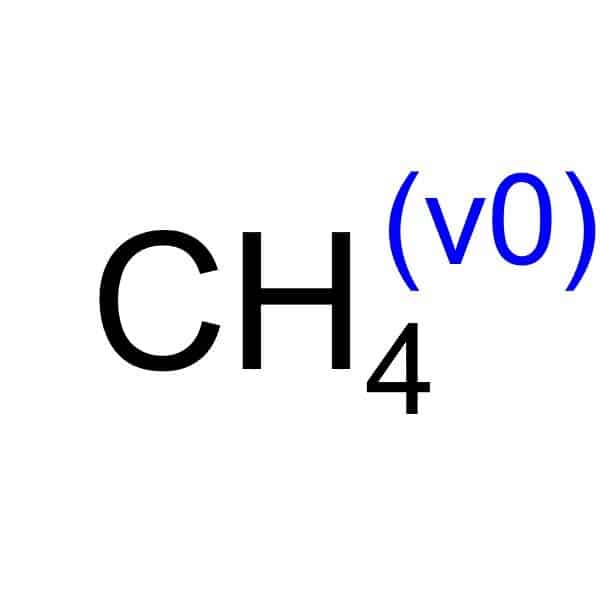
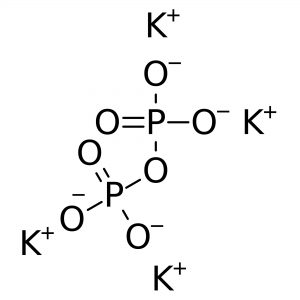
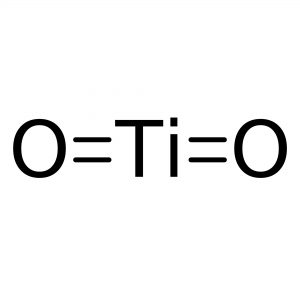
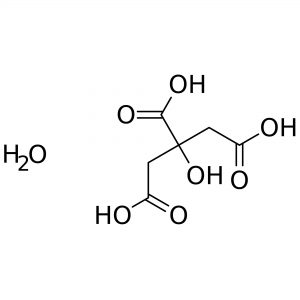
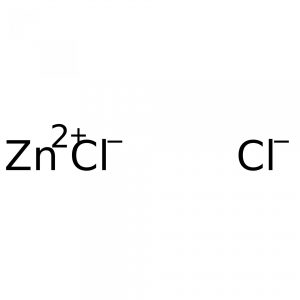
Reviews
There are no reviews yet.