Aluminum sulfate 17% [10043-01-3]
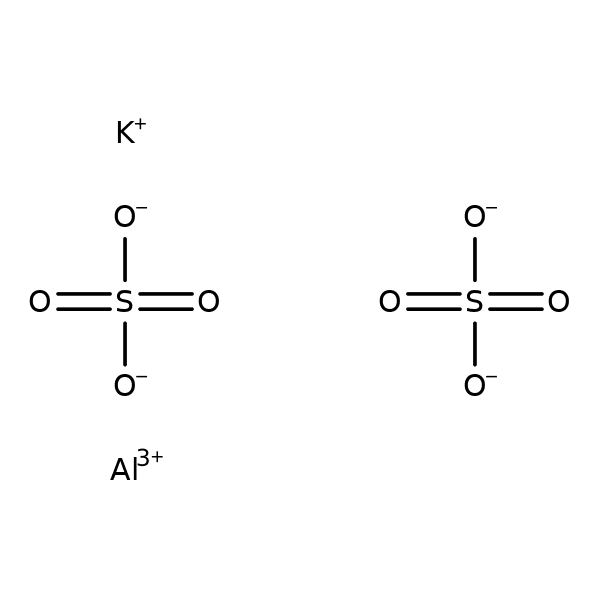
Aluminum Sulfate, an inorganic chemical compound, a salt of sulfuric acid and aluminum. Forms a series of hydrates, the most common of which are hexadecahydrate and octadecahydrate. Heptadecahydrate occurs naturally as the mineral alunogen. Hydrates contain six water molecules firmly bound as ligands of the aluminum atom, the remaining water molecules are part of the crystal lattice. Used as a dye mortar, in the paper industry, tanning and as an intermediate for the preparation of other aluminum compounds [4] [5]. It is an auxiliary food additive authorized for use in the European Union under the code E520 [6]. It is used for drinking water purification as a coagulating agent [7]. It has also found application in medicine. Used incl. in gynecology in the form of 1–4% aqueous solutions for vaginal irrigation.
Density:2,67 g/cm³
Melting point: 650 °C
Boiling point: 330 ° C
Solubility: In water 880 g / l
Molar mass: 342.15 g / mol
Formula: Al2 (SO4) 3
Hazard pictograms
Labeling of hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosive effect, the exclamation mark pictogram should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms




Reviews
There are no reviews yet.