Anhydrous potassium fluoride 99% AR [7789-23-3] Potassium fluoride is used in organic synthesis as a catalyst for various reactions or for introducing fluoride into organic molecules. For example, fluorine compounds can be produced by replacing labile chlorine atoms with fluorine atoms, such as in the production of sodium fluoroacetate, a rat poison. The F- nucleophilic strength and solubility of KF in aprotic organic solvents can be improved by using crown ethers.
Boiling point: 1505 ° C (1013 hPa)
Density: 2.48 g / cm3 (20 ° C)
Melting point: 846 ° C
PH value: 8 – 9 (50 g / l, H₂O, 20 ° C)
Vapor pressure: 1.3 hPa (885 ° C)
Bulk density: 400 kg / m3
Solubility: 923 g / l
Free bases (as KOH): ≤ 0.05%
Free acid (as HF): ≤ 0.05%
Chloride (Cl): ≤0.005%
Hexafluorosilicate (SiF₆): ≤0.05%
Sulphate (SO₄): ≤0.005%
Heavy metals (as Pb): ≤0.001%
Fe (iron): ≤0.001%
Na (sodium): ≤0.2%
Hazard pictograms
Labeling of hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosive effect, the exclamation mark pictogram should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms




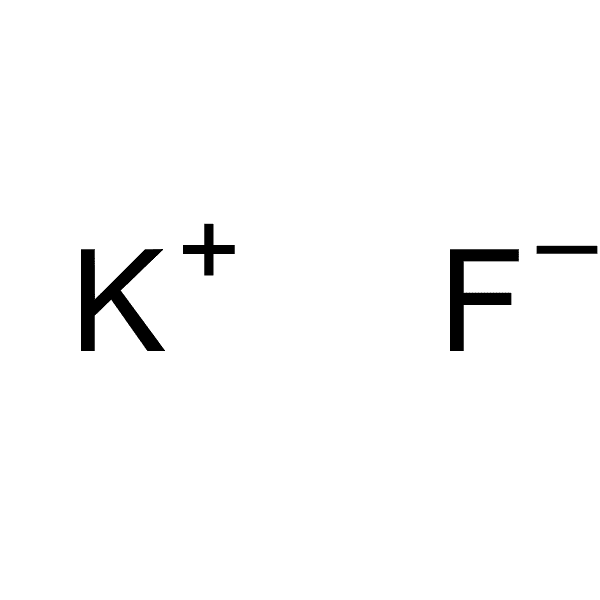
Reviews
There are no reviews yet.