Bromine 99,7 – 99,9% [7726-95-6]
Bromine is used in many reactions, e.g. for the synthesis of alkyl bromides, which are more reactive than the corresponding chlorides. Silver bromide has been used extensively in lithographic and photographic techniques.
General information:
Appearance Liquid
Color Red-brown
Odor Suffocating
Melting point -7,25 °C
Boiling point 58,8 – 59,5 °C
Flash point Not applicable
Ignition point Not applicable
Autoignition point Not applicable
Vapor pressure (at 20 oC) 173 mm Hg
Density (at 20 oC) 3,12 g/cm3
pH of aqueous extract (within
the range)
Not available
Solubility in/Miscibility with
water and other solvents
Soluble in water, chloroform, carbon tetrachloride, ethanol,
ether, carbon disulfide, concentrated hydrochloric acid.
Oxidizing properties Strong oxidizer
Solubility in other solvents: it dissolves in ethyl alcohol, ethyl ether, chloroform, carbon tetrachloride, carbon disulphide, acetone, nitrobenzene, in concentrated hydrochloric and hydrobromic acids, bromide solutions.
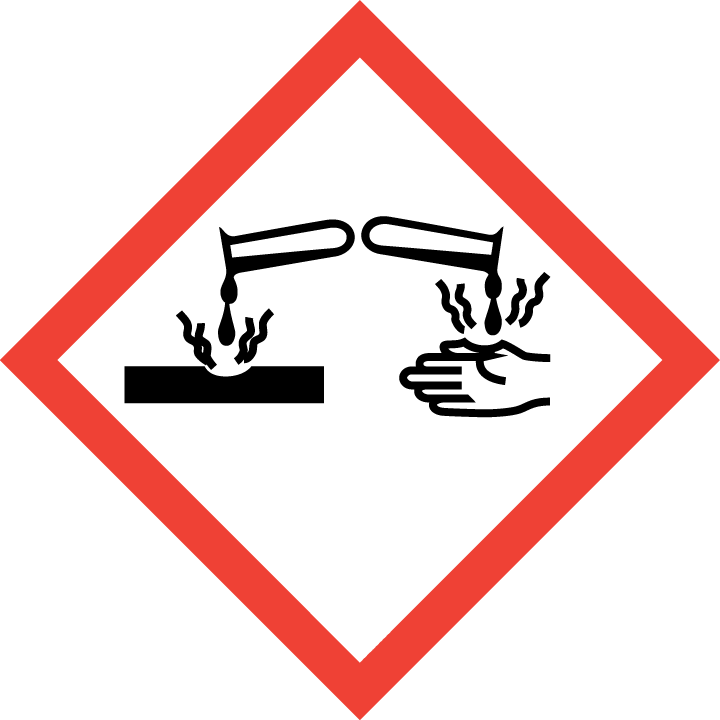
Hazard pictograms
Labeling of hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosive effect, the exclamation mark pictogram should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms








Reviews
There are no reviews yet.