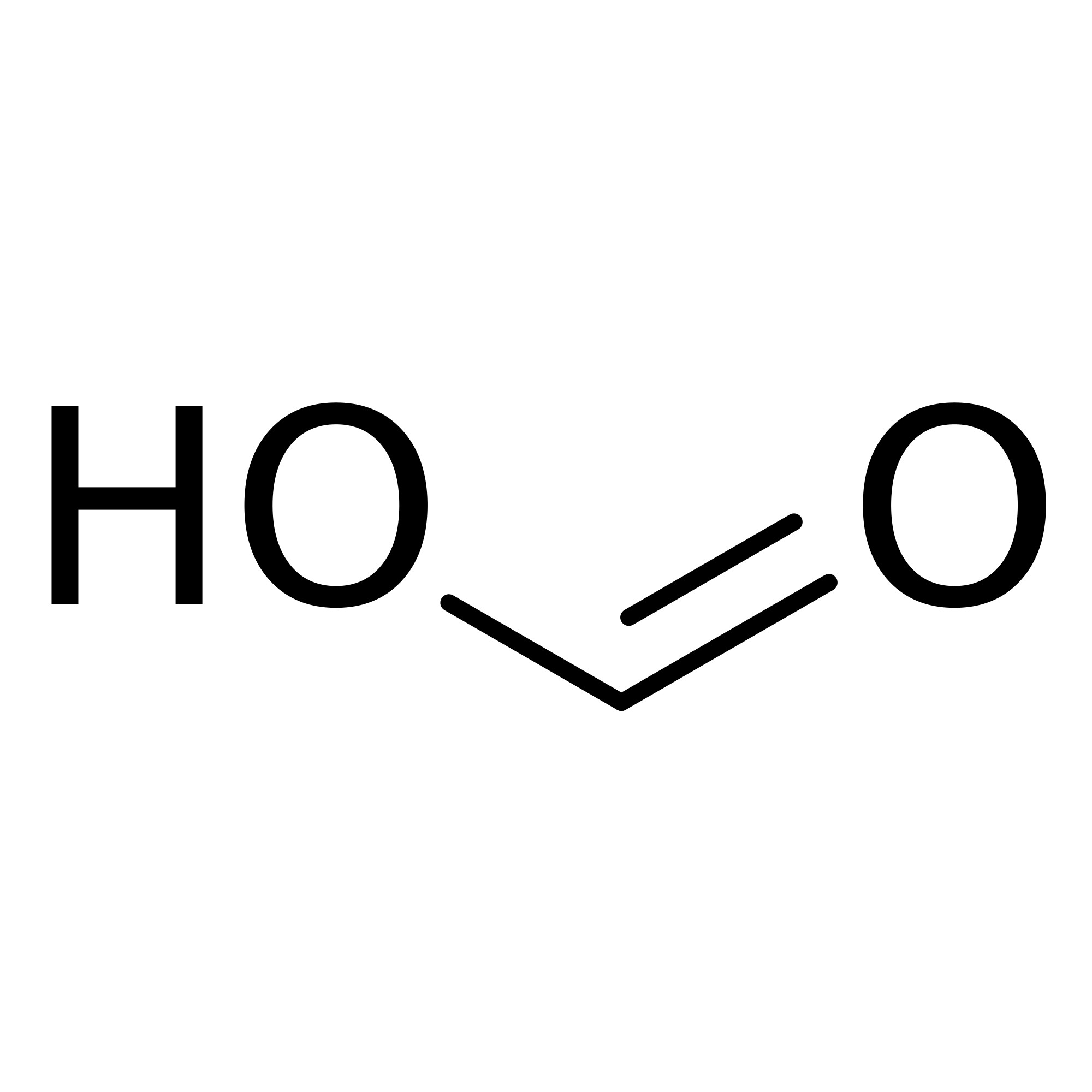
Formic acid [64-18-6]
Formic acid due to its fungicidal properties often used as a component of fungicidal and acidifying preparations. They often appear in combination with other acids or applied to a carrier. It is widely used in organic synthesis. In beekeeping, it is used to treat Varroa destructor mites. Formic acid is also used as a preservative and strong fungicidal properties, at high concentrations and pH ranges in its range of action also includes bacteria. As a food additive, it has the designation E236.
Physical state at 20C: liquid
Color: colorless
Aroma: sharp, penetrating
pH: strongly acidic
Melting point: – 8.4C
Boiling point: 100.7C
Flash point: 69C
Auto-ignition temperature: 434C
– lower: 18% vol.
– upper: 57% vol.
Stoichiometric concentration: 29.58% vol.
Density at 20C: 1.19-1.22 g / cm3
Vapor density against air: 1.6
Hazard pictograms
Labels for hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosivity, the pictogram exclamation mark should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms




Reviews
There are no reviews yet.