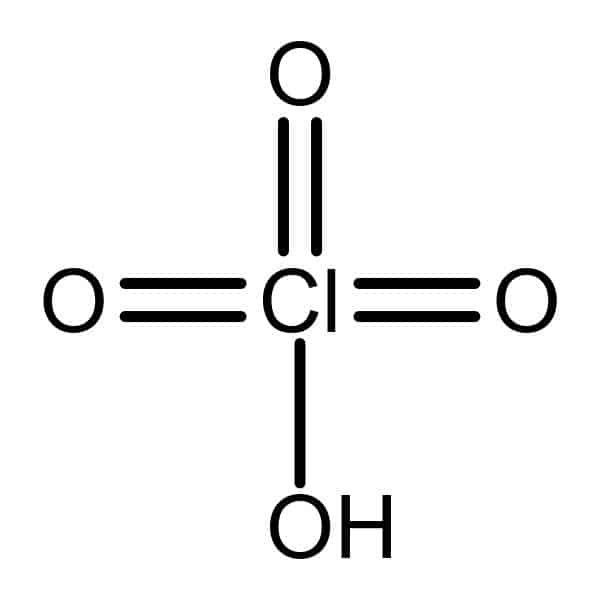
Perchloric acid 20% [7601-90-3] We use perchloric acid in analytical chemistry as an oxidant and to separate potassium from sodium. Its salts for explosives and for electroplating metals. Perchloric acid salts are used as explosives and for coating metals. They are also used as an anoxidizer and as a reagent in chemical analysis. These salts are prepared by distilling potassium chlorate with concentrated H2SO4 under reduced pressure.
Melting point: -18 ° C
Boiling point: 203 ° C
Density: 1.664 g / ml at 25 ° C
vapor density: ~ 2.1 (vs air)
vapor pressure: 6.8 mm Hg (25 ° C)
refractive index: 1.419
Flash point: 104 ° F
Specific gravity: about 1.54
Odor: odorless
PH: 0.1 (H2O, 20 ° C)
Hazard pictograms
Labeling of hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosive effect, the exclamation mark pictogram should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms




Reviews
There are no reviews yet.