Iron chloride anhydrous 97% [7705-08-0] Iron chloride has a depressive effect on barite and can be used in barite-celestite separation. He was also rated as a depressant when separating niobium with zirconium. In general, iron and iron compounds are not selective depressants and in many cases are harmful to the flotation of oxide and industrial minerals, such as anion flotation, fatty acids, iron complexes or iron oleate complexes.
Density: 2.89 g / cm3 (25 ° C)
Melting point: 306 ° C (decomposition)
PH value: 1 (200 g / l, H₂O, 20 ° C)
Vapor pressure: 1 hPa (20 ° C)
Bulk density: 1000 kg / m3
Solubility: 920 g / l Hydrolysis
Identity (Fe): pass the test
Identity (Cl): pass the test
Hazard pictograms
Labeling of hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosive effect, the exclamation mark pictogram should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms




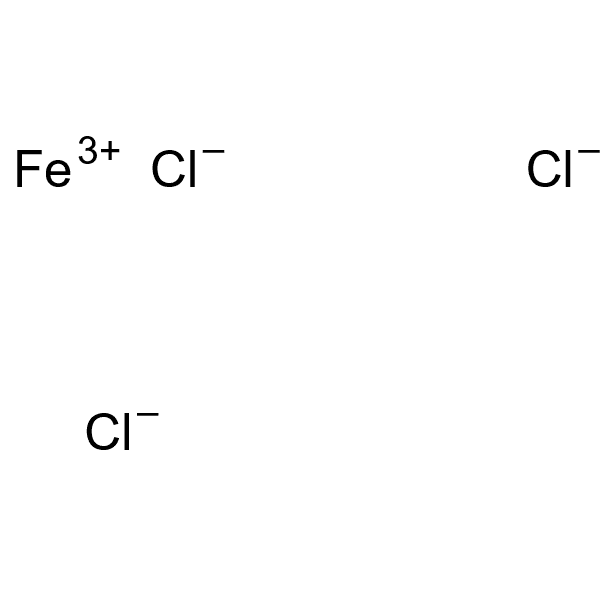
Reviews
There are no reviews yet.