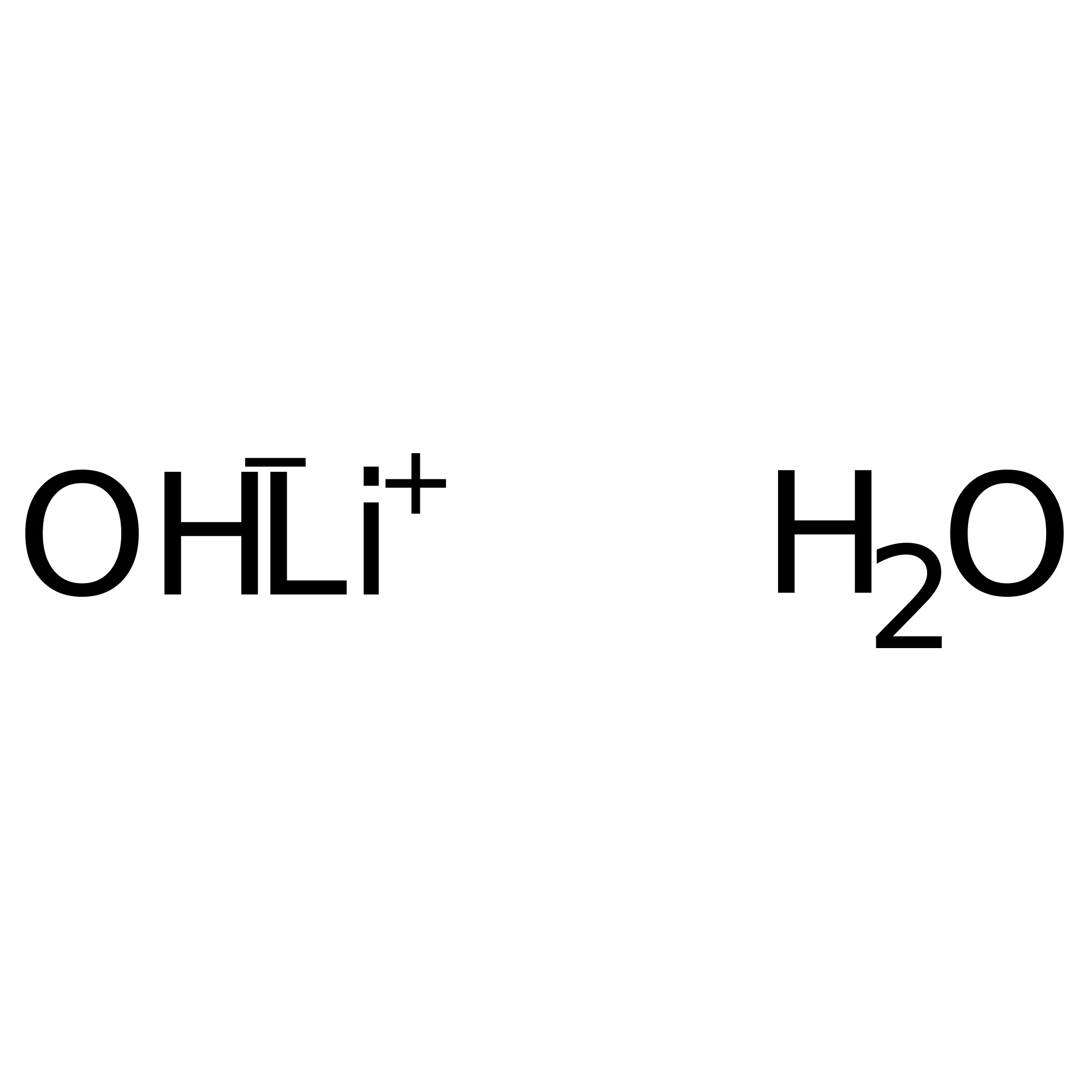Lithium hydroxide p.a. [1310-66-3]
The use of lithium hydroxide in industry: for the production of lithium greases, among others resistant to water and high and low temperatures – lithium stearate, for the production of lithium soap and lithium salts, in gas purification systems including for spacecraft, submarines and reactors, for CO2 removal, as an admixture for special bearing alloys, as a component improving the mechanical properties of aluminum and lead alloys, in nuclear technology, is part of the control rods in nuclear reactors, in organic synthesis as a catalyst in reactions esterification, in solution in aliphatic amines as a strong reducing agent, used in the production of alkaline batteries, accumulators (as an electrolyte), used as a heat transfer medium, in ceramics and some Portland cements, to alkalize the reactor coolant in pressurized reactors to control corrosion.
Form: solid
White color
Odor: no smell
pH:> 13 (1% solution)
Melting point: 424 ° C
Bulk weight: approx. 650 kg / m3
Solubility: in water: 189-223 g / l (20 ° C)
Hazard pictograms
Labels for hazardous chemicals and mixtures that are part of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The pictograms recommended by GHS have the shape of a square set on the top. They should contain a black symbol on a white background with a red border.
Priority rules to be observed in connection with the labeling of a substance:
– the skull and crossbones, the exclamation mark pictogram should not be added.
– corrosivity, the pictogram exclamation mark should not be added if it concerns eye or skin irritation.
– health hazard determining respiratory sensitization, the exclamation mark pictogram should not be added if it concerns skin sensitization or irritation to eyes or skin.
Source: GHS pictograms